The Growing Trend Of Clinical Research Crowdsourcing
 Margarita KitovaThe trend of open collaboration has led to innovation across multiple industries. For decades, big pharma has been known as conservative and slow to change. Today however, there is a growing movement toward open access and crowdsourcing scientific information to accelerate research and development. Open-source platforms have let developers create multiple crowdsourcing applications, that are further enabling the crowdsourcing trend in the life sciences industry, as well.
Margarita KitovaThe trend of open collaboration has led to innovation across multiple industries. For decades, big pharma has been known as conservative and slow to change. Today however, there is a growing movement toward open access and crowdsourcing scientific information to accelerate research and development. Open-source platforms have let developers create multiple crowdsourcing applications, that are further enabling the crowdsourcing trend in the life sciences industry, as well.
Apple recently announced its ResearchKit, an open source software framework for researchers that allows developers to design health and fitness apps that can communicate with each other. This open source framework can give researchers fast and easy access to more clinical data than ever before.
Many patients today no longer want to be passive recipients of medical care, just research subjects, or data providers. They want to become problem solvers who participate in a global community of stakeholders. Online physician communities are breaking down competitive barriers and collaborating with each other in order to provide better care for their patients.
A recent example comes from SERMO, a global social crowdsourcing network for physicians and medical providers whose members helped save a little girl’s life from a deadly e. coli strain that killed her brother [1]. Doctors from around the world worked seamlessly as a team across borders on this case, recommending testing and treatment, and reducing the risk of infection in her community.
Another great example of the power of crowdsourcing comes from Stanford University, where a group of researchers stumbled upon an unexpected discovery- while examining autopsied brain tissue of multiple sclerosis (MS) patients, they noticed high levels of angiotensin enzymes, responsible for hypertension. That is how they came up with the hypothesis that angiotensin inhibitor (Lisinopril) would decrease MS flare-ups, and when they used it on mice with ML-like nerve damage, it reversed their paralysis [2]. In 2012, the FDA approved Lisinopril as the first investigational new drug developed through crowdsourcing.
 SERMO e-coli infographic
SERMO e-coli infographic
Carol Cruz Morton, writing in an article on this story published in IEEE Pulse outlines:
Since its early days, the Internet has been a popular forum for sharing health information. But the rise of Web 2.0 technologies, allowing individuals to measure, collect, and share their own health data, is beginning to transform the closed and competitive world of biomedical innovation and clinical research. Crowdsourcing, the solicitation of ideas from a large group of people through the Internet, is increasingly being used as a means of conducting research. Proponents say it can be a quick, cheap, and effective way of gathering study participants, ideas, and data. Many observers believe the large groundswell of collective knowledge provided through crowdsourcing will ultimately provide key insights into diseases and help get new, better, affordable drugs and devices to the right patients faster.
Experts say this phenomenon has emerged from three main trends that are changing the health research environment:
- Citizen science (non-expert trained individuals conducting science-related activities);
- Crowdsourcing (using web-based technologies to recruit clinical trial participants); and
- Medicine 2.0 (active participation of individuals in their health care particularly using the Internet and social media) [3].
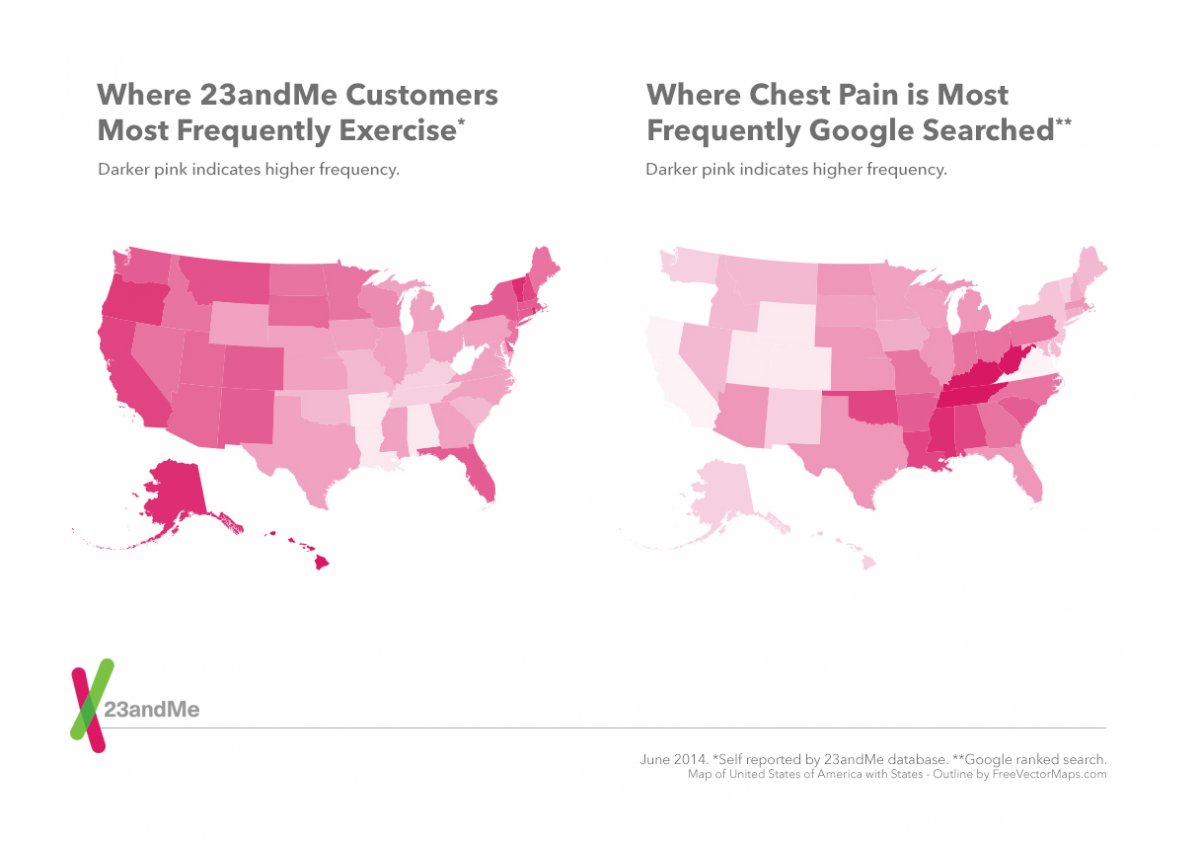
Among the leading health research crowdsourcing platforms are the personal genetics company 23andMe and the health information sharing website PatientsLikeMe. 23andMe has more than a million customers, 80% of which have allowed their DNA samples to be used in research. On a typical week, the company collects more than two million individual survey responses from its online community, contributing to over 230 research studies on the human genome [4].
PatientsLikeMe is the largest free network where patients with life-changing conditions can track and share their experiences to help each other, as well as researchers, pharmaceutical companies, regulators, providers and non-profits, develop better solutions to their problems. The network has a community of over 220,000 members who share their personal data on more than 2,000 medical conditions [5].
In addition, patients have also started to organize their own research studies with the help of health social networks and online communities such as Quantified Self, Genomera, and DIYgenomics [3]. Recently, some 200 crowdsourced studies have been added to the PubMed database [6].
Crowd data mining
According to the Oxford Bioinformatics Journal, many recent studies on the adverse effects of drugs are mining crowd data from search logs, Twitter, online patient forums, FDA reports as well as electronic health records (EHRs).
While traditional reporting of drug adverse effects is slow and does not capture all cases, web search patterns can provide early clues to adverse drug interactions. For example, a recent study explored the possibility of two drugs (paroxetine and pravastatin) interacting adversely and causing hyperglycemia. The researchers analyzed 82 million drug queries from 6 million web searches in 2010 [7]. Patients opted to share their IP addresses and search activities through a Microsoft add-on browser application. The results confirmed that patients who took both drugs at the same time were more likely to search for hyperglycemia-related terms compared to those who searched for only one of the drugs.
Another study used Google Trends (online tool which gives statistics on search term use) to observe the seasonality of sleep disordered breathing. Using search terms such as “snoring,” “sleep apnea,” and “snoring children,” researchers collected queries from a seven-year time span in both the United States and Australia [8]. Using regression analysis, researchers found that peak searches happened in the early spring and early winter season in both countries.
Active crowdsourcing
As opposed to crowd data mining, active crowdsourcing uses community challenges, labor markets, open forums or games to encourage participation in pharmaceutical clinical trials [6]. Labor market platforms such as the Amazon Mechanical Turk or Figure Eight allow researchers to design and submit crowdsourcing tasks or build entire sophisticated interfaces around a study in order to recruit participants.
A group of scientists recently evaluated the viability of using crowdsourcing for creating large data sets through the public health forums MedHelp and CureTogether. They conducted two different studies, one with 30 professional nurses recruited from oDesk (freelance labor market), and another with 50 crowd workers recruited from MTurk [9]. The results showed that the crowd-labeled data achieved an accuracy of 84% compared to 78% for the expert-labeled data.
Yet another form of active crowd sourcing are scientific games such as “Foldit,” a protein folding game with over half a million registered players, which has dramatically improved protein folding algorithms. Foldit’s goal is to predict the structure of a protein through puzzle-solving scenarios which would aid the creation of designing new proteins to combat disease-related proteins found in HIV/AIDS, cancer or Alzheimer’s, for example [10].
 Compact 37-Residue Marburg Virus Inhibitor Design Puzzle - Credit LociOiling from the Beta Folders, FoldItWhy crowdsource?
Compact 37-Residue Marburg Virus Inhibitor Design Puzzle - Credit LociOiling from the Beta Folders, FoldItWhy crowdsource?
Big pharma has only scratched the surface in terms of utilizing the potential for accelerating R&D though crowdsourcing. The Internet and social media have created a new public health ecosystem that spans across state borders. According to Google, the number of smart phone users globally was 1.75 billion in 2014, and is projected to increase to 2 billion by the end of 2015 [11]. Consumers are increasingly using smart phone applications and wearable technology to track their personal health and are also more willing to share their data with researchers. This story is detailed by the article How Wearable Technology Will Change Clinical Trials.
Reduce cost
Crowd sourcing offers an innovative approach to reducing the cost of conducting clinical trials. Labor markets such as MTurk, for example, offer many convenient features for accessing a large pool of workers for recruitment. The cost of designing and running a study using this platform is reportedly modest compared to hiring staff directly and custom designing the trial setup. However, it is important to note that the goal here is to not replace traditional expert-driven clinical trials but rather enhance data quality and quantity without increasing cost.
On the patient side of the equation, managing severe chronic illnesses can be very costly. Many of these serious conditions lack effective treatments, which is another incentive for patients to hack their diseases.
Increase participation in clinical trials
Crowdsourcing facilitates education about clinical trials and can thus significantly improve participation rates. In the States, only about 3% of cancer patients decide to enroll in cancer clinical trials [12]. Recent research shows than educating the public can dramatically increase the number of volunteers and accelerate trial accrual, which ultimately gets questions answered quicker [13].
Improve disease understanding
Crowd sourcing allows access to large cohorts and real-time data. The SERMO social media for doctors platform has 382,000 members- all of whom are verified physicians with current credentials [14]. A lot of community doctors don’t have experience with clinical trial protocols but strive to remain active in contributing to advancing research. Crowdsourcing, therefore, provides them with the perfect outlet where they can choose their level of participation and the time they’d like to dedicate, and still develop research ideas with peers from across the globe. Ultimately, this fosters innovation and generates new drugs and treatments that better meet patients’ needs.
Break down barriers
Language or cultural barriers can also pose problems when a diverse population cohort is needed in a clinical study [15]. If the content were made available on an open platform, doctors, caregivers and patients could collaborate. This collaboration can provide more culturally sensitive solutions to their problems.
For example, during a 2007 medical conference, a group of Italian researchers reported that lithium (used for bipolar disease) had shown to delay amyotrophic lateral sclerosis (ALS) progression [2]. Even before their paper was published officially, ALS patients had already used Google translate to share the paper abstract in English. Soon enough, patients collaborated on a group Google spreadsheet, tracking their own personal data, and comparing it to that of the researchers in Italy. About 160 patients tested lithium on themselves, which prompted PatientsLikeMe to build more tools for data structuring. Some of the patients even posted their personal experience on YouTube.
Accelerate research and development
Crowdsourcing challenges are providing a financial incentive to participants attract a fresh pool of perspectives that can bring along new solutions outside the clinical research community. This format for data collection is also beneficial as the problem solvers don’t have access to each other’s algorithms, which allows for a better assessment.
A great example of this is the DREAM-Phil Bowen ALS Prediction Prize4Life challenge. Containing nearly nine thousand patients, PRO-ACT was launched as an open access platform for researchers in December 2012.
In the Prize4Life crowdsourcing challenge, solvers were asked to use three months of individual patient level clinical trial information to predict that patient’s disease progression over the subsequent nine months [16]. The challenge resulted in 37 unique algorithms from which two winning entries were selected.
Ultimately, the winning algorithms outperformed a baseline model as well as ALS clinicians using the same data. The organizers estimated that using both winning algorithms in future trial designs could reduce the required number of patients by at least 20%. The challenge also identified several potential predictors of disease progression (including uric acid, creatinine and surprisingly, blood pressure), shedding light on ALS research and development [16].
Drawbacks to crowdsourcing
Crowdsourcing does come with certain disadvantages. For example, in the cases where clinical trial sponsors don’t use existent crowdsourcing platforms (such as Amazon’s M Turk), they need to create a website for crowd participation, which increases clinical trial cost. Some experts also criticize crowdsourcing initiatives, such as contests, for being unethical and rewarding a few individuals while the work of the majority is exploited for free. The two main clinical crowdsourcing drawbacks, however, are the quality of data and the unregulated nature of this new trend.
Noisy data
In crowd sourced clinical studies, the sheer volume of gathered data can cause clutter and make it hard to dig through the unstructured and noisy information. Another reported complaint is that crowdsourcing labor market platforms such as Amazon’s MTurk can’t weed out spammers or poorly performing workers.
On the employee side, there is no guarantee of payment for their work, which is why the federal government is reluctant to approve such research studies [17]. In addition, MTurk studies’ quality control and evaluation are said to be dependent on domain experts, which deems it unsuitable for complex studies with sophisticated algorithms. This is also not a viable option for clinical studies that contain sensitive information and thus rely on confidentiality.
Regulatory hurdles
Crowdsourcing is a new, uncharted territory, still unregulated by most governments. However, innovation calls for change and the social trend of sharing personal health and disease information has started to change the traditional circumstances around clinical research and development.
The American Health Information Management Association (AHIMA), calls this trend “biosociality” [18]. People tend to feel more comfortable sharing personal details on online health-related forums because of the interpersonal context. On the other hand, sharing information with clinical trial researchers means a much more formal setting. And while patients who share their self-tracked data expect anonymity, there is no consensus on privacy and ownership standards in the health data space.
A recent customer survey on data ownership shows that 57% of consumers would only share their data on the condition of privacy protection. Over 90% said this is very important to them, and a small percentage were open to sharing their data with the mobile device company that collected it [19].
The lack of industry standards around crowdsourcing also raises concern about the validity of the collected patient health data. However, experts believe that the mainstream adoption of tech health devices would force the industry to address information governance and impose standards in the near future.
Future of clinical crowdsourcing
New platforms, products and services leveraging the power of the crowd are constantly emerging on the market. Amateur scientists are looking for crowd funding to start their own clinical studies on platforms such as GoFundMe and IndieGogo. The wide adoption of smart technology and social media are prompting new behavioral boundaries where sharing personal health data is acceptable. Google X is working on a clinical trial wristband that would allow scientists to gather health data from thousands of users. "With more than 2 billion smartphones and advances in biosensors, crowdsourcing will benefit through the automatic collection of billions and billions of data about the human condition,” says Marc Desgrousilliers, Chief Technology Officer at Clinovo, an open source, technology focused, Contract Research Organization (CRO) headquartered in the Silicon Valley.
However, crowdsourcing is still very much a buzzword, and it is still in a growing, experimental stage. Many argue that it cannot be fully utilized in clinical research and development until the FDA regulates it. Even if this happens in the near future, experts agree that crowd sourced health research is not meant to replace traditional clinical studies but rather complement and extend them. It will be very interesting to see how it evolves over time as tools and apps become common.
Sources:
[1] Global Medical Crowdsourcing Helped Save Girl from Deadly E. Coli Strain That Killed Brother.
[4] 23andMeMedia
[5] PatientsLikeMe
[6] Crowdsourcing in biomedicine: challenges and opportunities.
[7] Web-scale pharmacovigilance: listening to signals from the crowd
[8] Seasonal trends in sleep-disordered breathing: evidence from Internet search engine query data.
[9] Identifying medical terms in patient-authored text: a crowdsourcing-based approach (CC)
[10] The Science Behind Foldit: Why is this game important?
[12] Why Are Only 3% of US Cancer Patients in Clinical Trials?
[14] What is SERMO?
[15] Three Ways to Improve Clinical Trials through Crowdsourcing
[16] Crowdsourced analysis of clinical trial data to predict amyotrophic lateral sclerosis progression
[18] AHIMA
[19] Information Privacy Concerns: Linking Individual Perceptions with Institutional Privacy Assurances
- Tags:
- 23andMe
- adverse drug interactions
- Amazon Mechanical Turk
- American Health Information Management Association (AHIMA)
- Apple ResearchKit
- biomedical innovation
- biosociality
- Carol Cruz Morton
- citizen science
- clinical data
- clinical research
- clinical trial participants
- Clinovo
- Contract Research Organization (CRO)
- crowdsourcing
- crowdsourcing applications
- crowdsourcing scientific information
- CureTogether
- DIYgenomics
- DREAM-Phil Bowen ALS Prediction Prize4Life challenge
- electronic health records (EHRs)
- Figure Eight
- Foldit
- Genomera
- GoFundMe
- Google Trends
- IEEE Pulse
- Indiegogo
- life sciences industry
- Marc Desgrousilliers
- Margarita Kitova
- MedHelp
- Medicine 2.0
- MTurk
- oDesk
- open access (OA)
- open collaboration
- Open Source Platforms
- open source software framework
- Oxford Bioinformatics Journal
- PatientsLikeMe
- pharmaceutical industry
- public health forums
- Quantified Self
- SERMO
- Stanford University
- Web 2.0 technologies
- Login to post comments